
CAMK2 alpha, Active
Recombinant full-length human CAMK2alpha was expressed in Sf9 cells using an N-terminal GST tag.
Catalog No. C11-10G
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| C11-10G-05 | 5 ug | $226 | |
| C11-10G-10 | 10 ug | $325 | |
| C11-10G-BULK | BULK | Contact Us |

Recombinant full-length human CAMK2alpha was expressed in Sf9 cells using an N-terminal GST tag.
Catalog No. C11-10G
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| C11-10G-05 | 5 ug | $226 | |
| C11-10G-10 | 10 ug | $325 | |
| C11-10G-BULK | BULK | Contact Us |
Overview:
CAMK2α is a ser/thr protein kinase that is a member of the Ca2+/calmodulin-dependent protein kinase family. CAMK2α is abundant in the brain as a major constituent of the postsynaptic density and is required for hippocampal long-term potentiation (LTP) and spatial learning. In addition to its Ca2+/calmodulin-dependent activity, CAMK2α can undergo autophosphorylation, resulting in Ca2+/calmodulin-independent activity. The protein level of CAMK2α fluctuates during neuronal activity in cultured rat pup hippocampal neurons. The levels of CAMK2α increased with heightened neuronal activity (2).
Gene Aliases:
CAMKA; KIAA0968
Genbank Number:
References:
1. Silva, A J. et al: Impaired spatial learning in alpha-calcium calmodulin kinase II mutant mice. Science 257: 206-211, 1992.
2. Thiagarajan, T C. et al: Alpha- and beta-CaMKII: inverse regulation by neuronal activity and opposing effects on synaptic strength. Neuron 36: 1103-1114, 2002.
Specific Activity:
Sample Kinase Activity Plot. For specific information on a given lot, see related technical data sheet.
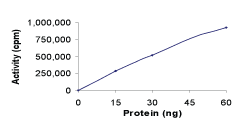
|
Purity:
Sample Purity Data. For specific information on a given lot, see related technical data sheet.

|
Storage, Stability and Shipping:
Store product at –70oC. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Molecular Weight:
~74 kDa
![]() A Thorne Curtis et al., Small-molecule inhibition of Wnt signaling through activation of casein kinase 1α Nature Chemical Biology November 2010 10.1038/nchembio.453
A Thorne Curtis et al., Small-molecule inhibition of Wnt signaling through activation of casein kinase 1α Nature Chemical Biology November 2010 10.1038/nchembio.453
![]() Caroccia Alessia et al., N-(Phenoxyalkyl)amides as MT(1) and MT(2) ligands: antioxidant properties and inhibition of Ca(2+)/CaM-dependent kinase II. Bioorganic & Medicinal Chemistry February 2013 10.1016/j.bmc.2012.12.017
Caroccia Alessia et al., N-(Phenoxyalkyl)amides as MT(1) and MT(2) ligands: antioxidant properties and inhibition of Ca(2+)/CaM-dependent kinase II. Bioorganic & Medicinal Chemistry February 2013 10.1016/j.bmc.2012.12.017
![]() Cipolletta E et al., Calmodulin-Dependent Kinase II Mediates Vascular Smooth Muscle Cell Proliferation and Is Potentiated by Extracellular Signal Regulated Kinase Endocrinology June 2010 10.1210/en.2009-1248
Cipolletta E et al., Calmodulin-Dependent Kinase II Mediates Vascular Smooth Muscle Cell Proliferation and Is Potentiated by Extracellular Signal Regulated Kinase Endocrinology June 2010 10.1210/en.2009-1248
![]() A. Witczak Carol et al., CaMKII regulates contraction- but not insulin-induced glucose uptake in mouse skeletal muscle. American Journal of Physiology - Endocrinology and Metabolism June 2010 10.1152/ajpendo.00659.2009
A. Witczak Carol et al., CaMKII regulates contraction- but not insulin-induced glucose uptake in mouse skeletal muscle. American Journal of Physiology - Endocrinology and Metabolism June 2010 10.1152/ajpendo.00659.2009
![]() Illario Maddalena et al., Calcium-calmodulin-dependent kinase II (CaMKII) mediates insulin-stimulated proliferation and glucose uptake Cellular signalling January 2007 10.1016/j.cellsig.2009.01.022
Illario Maddalena et al., Calcium-calmodulin-dependent kinase II (CaMKII) mediates insulin-stimulated proliferation and glucose uptake Cellular signalling January 2007 10.1016/j.cellsig.2009.01.022
![]() C Bruno et al., The chemosensitizing agent lubeluzole binds calmodulin and inhibits Ca(2+)/calmodulin-dependent kinase II. European Journal of Medicinal Chemistry June 2016 10.1016/j.ejmech.2016.03.045
C Bruno et al., The chemosensitizing agent lubeluzole binds calmodulin and inhibits Ca(2+)/calmodulin-dependent kinase II. European Journal of Medicinal Chemistry June 2016 10.1016/j.ejmech.2016.03.045
![]() Monterrey Gomez et al., Characterization of a selective CaMKII peptide inhibitor European Journal of Medicinal Chemistry April 2013 10.1016/j.ejmech.2012.12.053
Monterrey Gomez et al., Characterization of a selective CaMKII peptide inhibitor European Journal of Medicinal Chemistry April 2013 10.1016/j.ejmech.2012.12.053
![]() M Tandon et al., SD-208, a novel protein kinase D inhibitor, blocks prostate cancer cell proliferation and tumor growth in vivo by inducing G2/M cell cycle arrest. PLoS One March 2015 10.1371/journal.pone.0119346
M Tandon et al., SD-208, a novel protein kinase D inhibitor, blocks prostate cancer cell proliferation and tumor growth in vivo by inducing G2/M cell cycle arrest. PLoS One March 2015 10.1371/journal.pone.0119346
![]() Tandon Manuj et al., New Pyrazolopyrimidine Inhibitors of Protein Kinase D as Potent Anticancer Agents for Prostate Cancer Cells PLoS One September 2013 10.1371/journal.pone.0075601
Tandon Manuj et al., New Pyrazolopyrimidine Inhibitors of Protein Kinase D as Potent Anticancer Agents for Prostate Cancer Cells PLoS One September 2013 10.1371/journal.pone.0075601
![]() Tandon Manuj et al., A Targeted Library Screen Reveals a New Inhibitor Scaffold for Protein Kinase D PLoS One September 2012 10.1371/journal.pone.0044653
Tandon Manuj et al., A Targeted Library Screen Reveals a New Inhibitor Scaffold for Protein Kinase D PLoS One September 2012 10.1371/journal.pone.0044653
Cardiovascular Disease, ERK/MAPK Pathway, Neurobiology, PKA/PKC Pathway, Ser/Thr Kinases

 CAMK1 beta, Active, C08-10G
CAMK1 beta, Active, C08-10G

 CAMK4, Active, C15-10G
CAMK4, Active, C15-10G

 CAMK2 beta, Active, C12-10H
CAMK2 beta, Active, C12-10H

 CAMK2 delta, Active, C13-10G
CAMK2 delta, Active, C13-10G
STAY CONNECTED
Fax: 1-604-232-4601