Arginine Deiminase and Proteins
Arginine Deiminase and Proteins
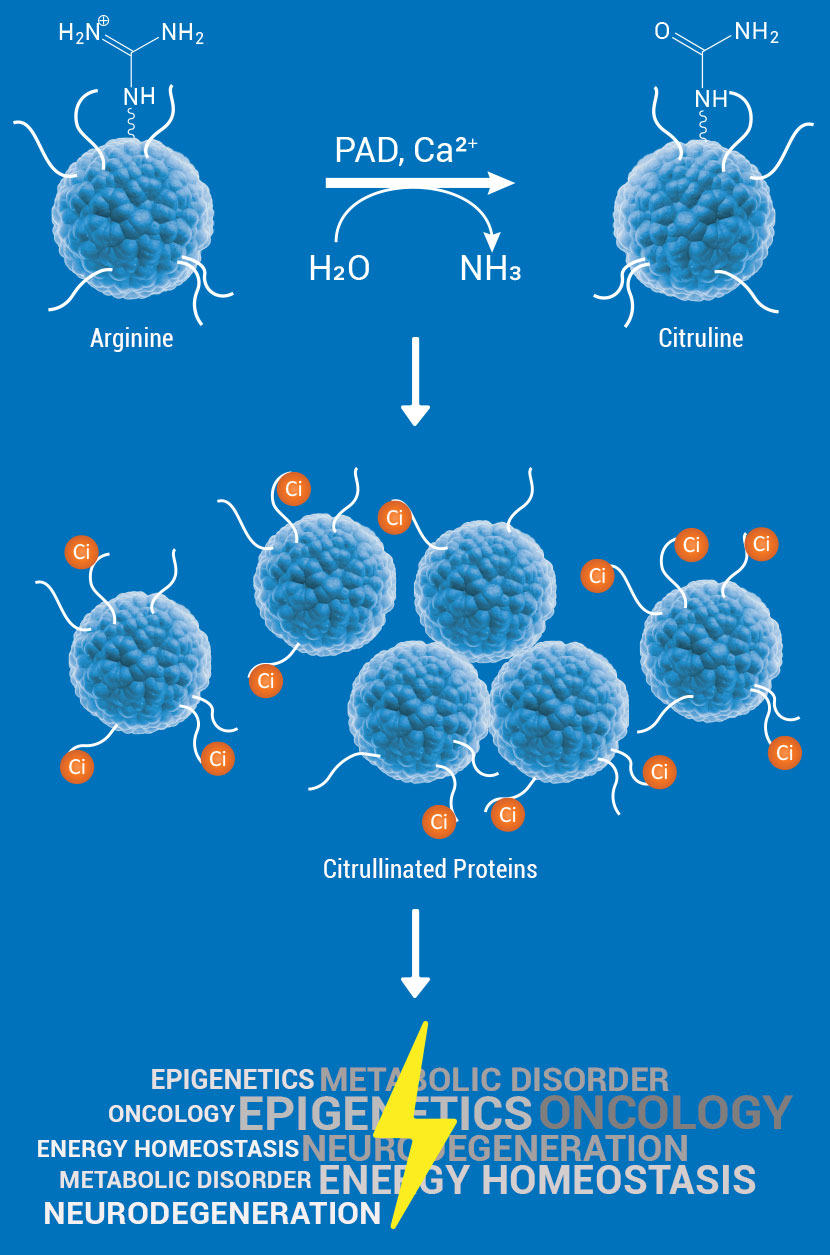
Post-translational modification of cellular proteins plays a crucial role in cell survival, cell signaling and overall maintenance of cellular health. Citrullination is one of the many crucial protein modifications catalyzed by a family of enzymes called Protein Arginine Deiminases (PADs). PADs catalyze the hydrolysis of peptidyl-arginine to form peptidyl-citrulline on important cellular proteins such as histones, thereby regulating multiple cell signaling pathways, and biochemical processes.
The PAD family consists of five members of calcium-dependent isozymes PAD1, 2, 3, 4, and 6. Citrullination or hypercitrullination of proteins induces a structural change, modifying their biological function and their interaction mechanisms with other cellular proteins. Because of this property, numerous notable studies have established PADs as an important class of targets for drug discovery projects related to many human diseases such as cancer, autoimmune diseases (rheumatoid arthritis), and neurodegeneration.
SignalChem’s scientists have produced all five high-quality recombinant mammalian PADs and have developed sensitive and reliable PAD assay cocktails to facilitate your drug discovery and development research. These are effective in vitro tools to investigate protein citrullination in various human diseases.

 Toll Free: 1-866-954-6273
Toll Free: 1-866-954-6273 info@signalchem.com
info@signalchem.com