
Anti-phospho-GSK3 beta (Ser9)
Rabbit Polyclonal Antibody
Catalog No. G09-65R
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| G09-65R-100 | 100 ug | $325 | |
| G09-65R-BULK | BULK | Contact Us |

Rabbit Polyclonal Antibody
Catalog No. G09-65R
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| G09-65R-100 | 100 ug | $325 | |
| G09-65R-BULK | BULK | Contact Us |
Overview:
STAT3 is a member of the signal transducers and activators of transcription (STAT) family of proteins that carry out dual functions. STAT3 is a DNA binding protein that is widely expressed and becomes activated upon phosphorylation on Tyr708, which leads dimerization, intracellular translocation and DNA-binding. Phosphorylation at Ser727 regulates transcriptional activity. STAT3 is activated in response to variety of stimuli, which includes EGF, IL-6, PDGF, IL-2 and G-CSF (1). STAT3 forms homo- and hetero-dimers with other STAT proteins to modulate the transcription of various genes (2).
References:
1. Zhong, Z. et al: Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science. 1994 Apr 1;264(5155):95-8.
2. Tian, S. S. et al: Rapid activation of the STAT3 transcription factor by granulocyte colony-stimulating factor. Blood. 1994 Sep 15;84(6):1760.
Specificity:
Recognizes the GSK3 protein phosphorylated at serine 9
Cross Reactivity:
Human, Mouse and Rat
Host / Isotype / Clone#:
Rabbit, IgG
Immunogen:
Synthetic phospho-peptide corresponding to amino acid residues surrounding Ser9
Purification:
Affinity chromatography
Stability:
Store at 4oC (add 0.1% NaN3) for several months, and at -20oC for longer periods. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Sample Data:
Western blots from HeLa cells using anti-phospho- phospho-GSK3 beta (Ser9) antibody.

|
Sample Data:
Immunohistochemical analysis of paraffin-embedded human breast carcinoma tissue using anti-phospho-GSK3 beta (Ser9) antibody.
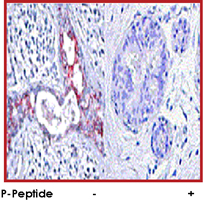
|
![]() Ishizuka Koko et al., DISC1-dependent switch from progenitor proliferation to migration in the developing cortex Nature April 2011 10.1038/nature09859
Ishizuka Koko et al., DISC1-dependent switch from progenitor proliferation to migration in the developing cortex Nature April 2011 10.1038/nature09859
![]() Tang Wenwen et al., A PLCβ/PI3Kγ-GSK3 Signaling Pathway Regulates Cofilin Phosphatase Slingshot2 and Neutrophil Polarization and Chemotaxis Developmental Cell December 2011 10.1016/j.devcel.2011.10.023
Tang Wenwen et al., A PLCβ/PI3Kγ-GSK3 Signaling Pathway Regulates Cofilin Phosphatase Slingshot2 and Neutrophil Polarization and Chemotaxis Developmental Cell December 2011 10.1016/j.devcel.2011.10.023
![]() I Pronobis Mira et al., A novel GSK3-regulated APC:Axin interaction regulates Wnt signaling by driving a catalytic cycle of efficient ?catenin destruction Elife September 2015 10.7554/eLife.08022
I Pronobis Mira et al., A novel GSK3-regulated APC:Axin interaction regulates Wnt signaling by driving a catalytic cycle of efficient ?catenin destruction Elife September 2015 10.7554/eLife.08022
![]() Li Saiqun et al., GSK3 Temporally Regulates Neurogenin 2 Proneural Activity in the Neocortex Journal of Neuroscience November 2012 10.1523/JNEUROSCI.1309-12.2012
Li Saiqun et al., GSK3 Temporally Regulates Neurogenin 2 Proneural Activity in the Neocortex Journal of Neuroscience November 2012 10.1523/JNEUROSCI.1309-12.2012
![]() FJ Gao et al., GSK-3β Phosphorylation of Cytoplasmic Dynein Reduces Ndel1 Binding to Intermediate Chains and Alters Dynein Motility. Traffic September 2015 10.1111/tra.12304
FJ Gao et al., GSK-3β Phosphorylation of Cytoplasmic Dynein Reduces Ndel1 Binding to Intermediate Chains and Alters Dynein Motility. Traffic September 2015 10.1111/tra.12304
![]() JN Kong et al., Regulation of Chlamydomonas flagella and ependymal cell motile cilia by ceramide-mediated translocation of GSK3. Molecular Biology of the Cell December 2015 10.1091/mbc.E15-06-0371
JN Kong et al., Regulation of Chlamydomonas flagella and ependymal cell motile cilia by ceramide-mediated translocation of GSK3. Molecular Biology of the Cell December 2015 10.1091/mbc.E15-06-0371
![]() S. Desai Shruti et al., GSK-3β Protein Phosphorylates and Stabilizes HLXB9 Protein in Insulinoma Cells to Form a Targetable Mechanism of Controlling Insulinoma Cell Proliferation Journal of Biological Chemistry January 2014 10.1074/jbc.M113.533612
S. Desai Shruti et al., GSK-3β Protein Phosphorylates and Stabilizes HLXB9 Protein in Insulinoma Cells to Form a Targetable Mechanism of Controlling Insulinoma Cell Proliferation Journal of Biological Chemistry January 2014 10.1074/jbc.M113.533612
![]() Yamaguchi Fuminori et al., S100 Proteins Modulate Protein Phosphatase 5 FunctionA LINK BETWEEN CA2+ SIGNAL TRANSDUCTION AND PROTEIN DEPHOSPHORYLATION Journal of Biological Chemistry April 2012 10.1074/jbc.M111.329771
Yamaguchi Fuminori et al., S100 Proteins Modulate Protein Phosphatase 5 FunctionA LINK BETWEEN CA2+ SIGNAL TRANSDUCTION AND PROTEIN DEPHOSPHORYLATION Journal of Biological Chemistry April 2012 10.1074/jbc.M111.329771
![]() Banerjee Sami et al., Modulation of SCFβ-TrCP-dependent IKBα Ubiquitination by Hydrogen Peroxide Journal of Biological Chemistry January 2010 10.1074/jbc.M109.060822
Banerjee Sami et al., Modulation of SCFβ-TrCP-dependent IKBα Ubiquitination by Hydrogen Peroxide Journal of Biological Chemistry January 2010 10.1074/jbc.M109.060822
![]() D Xu et al., Obg-like ATPase 1 regulates global protein serine/threonine phosphorylation in cancer cells by suppressing the GSK3β-inhibitor 2-PP1 positive feedback loop. Oncotarget January 2016 10.18632/oncotarget.6496
D Xu et al., Obg-like ATPase 1 regulates global protein serine/threonine phosphorylation in cancer cells by suppressing the GSK3β-inhibitor 2-PP1 positive feedback loop. Oncotarget January 2016 10.18632/oncotarget.6496
![]() Martic Sanela et al., Electrochemical Investigations of Tau Protein Phosphorylations and Interactions with Pin1 Chemistry & Biodiversity September 2012 10.1002/cbdv.201100418
Martic Sanela et al., Electrochemical Investigations of Tau Protein Phosphorylations and Interactions with Pin1 Chemistry & Biodiversity September 2012 10.1002/cbdv.201100418
![]() Villanueva JO Esteves et al., Electrochemical detection of anti-tau antibodies binding to tau protein and inhibition of GSK-3β-catalyzed phosphorylation. Analytical Biochemistry March 2016 10.1016/j.ab.2015.12.002
Villanueva JO Esteves et al., Electrochemical detection of anti-tau antibodies binding to tau protein and inhibition of GSK-3β-catalyzed phosphorylation. Analytical Biochemistry March 2016 10.1016/j.ab.2015.12.002
![]() S. Darshita B et al., Identification and in vitro evaluation of new leads as selective and competitive glycogen synthase kinase-3β inhibitors through ligand and structure based drug design Journal of Molecular Graphics and Modelling July 2014 10.1016/j.jmgm.2014.06.013
S. Darshita B et al., Identification and in vitro evaluation of new leads as selective and competitive glycogen synthase kinase-3β inhibitors through ligand and structure based drug design Journal of Molecular Graphics and Modelling July 2014 10.1016/j.jmgm.2014.06.013
AKT/PKB Pathway, Apoptosis/Autophagy, Cancer, Cardiovascular Disease, Inflammation, Invasion/Metastasis, Metabolic Disorder, Neurobiology, NfkB Pathway, Ser/Thr Kinases, WNT Signaling
STAY CONNECTED
Fax: 1-604-232-4601