
Anti-phospho-ERK1/2 (Thr202/Tyr204)
Rabbit Polyclonal Antibody
Catalog No. M29-652R
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| M29-652R-100 | 100 ul | $715 | |
| M29-652R-BULK | BULK | Contact Us |

Rabbit Polyclonal Antibody
Catalog No. M29-652R
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| M29-652R-100 | 100 ul | $715 | |
| M29-652R-BULK | BULK | Contact Us |
Overview:
Extracellular-Signal Regulated Kinase/Mitogen-Activated Protein Kinase (ERK/MAPK) is a serine threonine kinase. It plays an integral role of cellular signaling during mitogenesis and differentiation of mitotic cells. ERK is presumed to have a key role in learning and memory (1,2,3). The activity of this kinase is regulated by phosphorylation at Thr202 and Tyr204 (4). Activated ERK1/2 translocates into the nucleus where it phosphorylates various transcription factors (e.g Elk-1, c-Myc, c-Jun, c-Fos and C/EBP beta).
References:
1. Adams, J P. et al: Molecular psychology: Roles for the ERK MAP kinase cascade in memory. Annu Rev Pharmacol Toxicol 2002 42:135-163.
2. Johnson, G L. et al: Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 202 298:1911-1912.
3. Tanoue, T J. et al: Molecular recognitions in the MAP kinase cascades. Cellular Signaling 2003 15:455-462.
4. Ahn, N G.: The MAP kinase cascade. Discovery of a new signal transduction pathway. Mol Cell Biochem 1993 127-128:201-209.
Specificity:
Recognizes the ERK1/2 protein phosphorylated at threonine 202 and tryrosine 204
Cross Reactivity:
Human, Mouse, Rat, Bovine, Canine, Chicken, non-Human Primate, Xenopus and Zebrafish
Host:
Rabbit, IgG
Immunogen:
Synthetic phospho-peptide corresponding to amino acid residues surrounding Thr202/Tyr204 conjugated to KLH
Purification:
Affinity Chromatography
Stability:
Store at 4oC (add 0.1% NaN3) for several months, and at -20oC for longer periods. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Sample Data:
Western blot of human T47D cell lysates showing specific immunolabeling of ~42-44kDa ERK1/2 protein phosphorylated at Thr202/Tyr204 (Control). Phosphospecificity is shown in the second lane (lambda-phosphatase: lambda-Ptase). The blot is identical to the control except that it was incubated in lambda-Ptase (1200 units for 30 min) before being exposed to the Anti-phospho-ERK1/2 (Thr202/Tyr204) antibody. The immunolabeling is completely eliminated by treatment with lambda-Ptase.
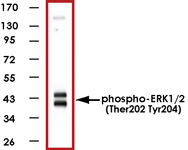
|
Sample Data:
Immunostaining of granule cells in the dentate gyrus of saline treated mouse showing ERK1/2 when phosphorylated at Thr202/Tyr204 (red) and nuclei (blue). Photo courtesy of Robert Wine.
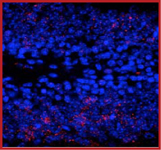
|
![]() Li Saiqun et al., RAS/ERK Signaling Controls Proneural Genetic Programs in Cortical Development and Gliomagenesis Journal of Neuroscience February 2014 10.1523/JNEUROSCI.4077-13.2014
Li Saiqun et al., RAS/ERK Signaling Controls Proneural Genetic Programs in Cortical Development and Gliomagenesis Journal of Neuroscience February 2014 10.1523/JNEUROSCI.4077-13.2014
![]() Tonga Junfeng et al., Phosphorylation of Rac1 T108 by Extracellular Signal-Regulated Kinase in Response to Epidermal Growth Factor: a Novel Mechanism To Regulate Rac1 Function Molecular and Cellular Biology November 2013 10.1128/MCB.00822-13
Tonga Junfeng et al., Phosphorylation of Rac1 T108 by Extracellular Signal-Regulated Kinase in Response to Epidermal Growth Factor: a Novel Mechanism To Regulate Rac1 Function Molecular and Cellular Biology November 2013 10.1128/MCB.00822-13
![]() E. Franklin Norah et al., Differential phosphorylation of the phosphoinositide 3-phosphatase MTMR2 regulates its association with early endosomal subtypes Journal of Cell Science February 2013 10.1242/jcs.113928
E. Franklin Norah et al., Differential phosphorylation of the phosphoinositide 3-phosphatase MTMR2 regulates its association with early endosomal subtypes Journal of Cell Science February 2013 10.1242/jcs.113928
![]() Tyler Moore et al., IRF3 and ERK MAP-kinases control nitric oxide production from macrophages in response to poly-I:C FEBS Letters September 2013 10.1016/j.febslet.2013.07.025
Tyler Moore et al., IRF3 and ERK MAP-kinases control nitric oxide production from macrophages in response to poly-I:C FEBS Letters September 2013 10.1016/j.febslet.2013.07.025
![]() Yanga Shuping et al., Phosphorylation of KIBRA by the extracellular signal-regulated kinase (ERK)?ribosomal S6 kinase (RSK) cascade modulates cell proliferation and migration Cellular Signaling February 2014 10.1016/j.cellsig.2013.11.012
Yanga Shuping et al., Phosphorylation of KIBRA by the extracellular signal-regulated kinase (ERK)?ribosomal S6 kinase (RSK) cascade modulates cell proliferation and migration Cellular Signaling February 2014 10.1016/j.cellsig.2013.11.012
![]() Neise Denise et al., Evidence for a differential modulation of p53-phosphorylating kinases by the cyclin-dependent kinase inhibitor p21WAFI/C1P1 Cell Cycle September 2010 10.4161/cc.9.17.12799
Neise Denise et al., Evidence for a differential modulation of p53-phosphorylating kinases by the cyclin-dependent kinase inhibitor p21WAFI/C1P1 Cell Cycle September 2010 10.4161/cc.9.17.12799
![]() AL Lasek et al., The Functional Significance of Posttranslational Modifications on Polo-Like Kinase 1 Revealed by Chemical Genetic Complementation. PLoS One February 2016 10.1371/journal.pone.0150225
AL Lasek et al., The Functional Significance of Posttranslational Modifications on Polo-Like Kinase 1 Revealed by Chemical Genetic Complementation. PLoS One February 2016 10.1371/journal.pone.0150225
![]() Tong Junfeng et al., Phosphorylation and Activation of RhoA by ERK in Response to Epidermal Growth Factor Stimulation PLoS One January 2016 10.1371/journal.pone.0147103
Tong Junfeng et al., Phosphorylation and Activation of RhoA by ERK in Response to Epidermal Growth Factor Stimulation PLoS One January 2016 10.1371/journal.pone.0147103
![]() N Sato et al., MEK and PI3K catalytic activity as predictor of the response to molecularity targeted agents in triple-negative breast cancer Biochemical and Biophysical Research Communications August 2017
N Sato et al., MEK and PI3K catalytic activity as predictor of the response to molecularity targeted agents in triple-negative breast cancer Biochemical and Biophysical Research Communications August 2017
![]() Bhandaria Deepali et al., Cyclin-dependent kinase 5 activates guanine nucleotide exchange factor GIV/Girdin to orchestrate migration?proliferation dichotomy PNAS July 2015 10.1073/pnas.1514157112
Bhandaria Deepali et al., Cyclin-dependent kinase 5 activates guanine nucleotide exchange factor GIV/Girdin to orchestrate migration?proliferation dichotomy PNAS July 2015 10.1073/pnas.1514157112
![]() R Pal et al., Inhibition of ERK1/2 Restores GSK3β Activity and Protein Synthesis Levels in a Model of Tuberous Sclerosis Sci Rep June 2017 10.1038/s41598-017-04528-5
R Pal et al., Inhibition of ERK1/2 Restores GSK3β Activity and Protein Synthesis Levels in a Model of Tuberous Sclerosis Sci Rep June 2017 10.1038/s41598-017-04528-5
![]() R Pal et al., Inhibition of ERK1/2 Restores GSK3β Activity and Protein Synthesis Levels in a Model of Tuberous Sclerosis Sci Rep June 2017 10.1038/s41598-017-04528-5
R Pal et al., Inhibition of ERK1/2 Restores GSK3β Activity and Protein Synthesis Levels in a Model of Tuberous Sclerosis Sci Rep June 2017 10.1038/s41598-017-04528-5
![]() K Fujita et al., HMGB1, a pathogenic molecule that induces neurite degeneration via TLR4-MARCKS, is a potential therapeutic target for Alzheimer's disease. Science Reports August 2016 10.1038/srep31895
K Fujita et al., HMGB1, a pathogenic molecule that induces neurite degeneration via TLR4-MARCKS, is a potential therapeutic target for Alzheimer's disease. Science Reports August 2016 10.1038/srep31895
Angiogenesis, Apoptosis/Autophagy, Cancer, Cardiovascular Disease, ERK/MAPK Pathway, Invasion/Metastasis, Neurobiology, Ser/Thr Kinases
STAY CONNECTED
Fax: 1-604-232-4601