
ERK1, Active
Recombinant full-length human ERK1 was expressed in E.coli using an N-terminal GST tag.
Catalog No. M29-10G
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| M29-10G-05 | 5 ug | $226 | |
| M29-10G-10 | 10 ug | $325 | |
| M29-10G-BULK | BULK | Contact Us |

Recombinant full-length human ERK1 was expressed in E.coli using an N-terminal GST tag.
Catalog No. M29-10G
| Catalog No. | Pack Size | Price (USD) | |
|---|---|---|---|
| M29-10G-05 | 5 ug | $226 | |
| M29-10G-10 | 10 ug | $325 | |
| M29-10G-BULK | BULK | Contact Us |
Overview:
ERK1 is a protein serine/threonine kinase that is a member of the extracellular signal-regulated kinases (ERKs), also known as mitogen-activated protein kinase 3 (MAPK3) which are activated in response to numerous growth factors and cytokines. ERK1 is ubiquitously distributed in tissues with the highest expression in heart, brain and spinal cord. Activated ERK1 translocates into the nucleus where it phosphorylates various transcription factors (e.g., Elk-1, c-Myc, c-Jun, c-Fos, and C/EBP beta). ERK pathway is necessary for experience-dependent plasticity and for long-term potentiation of synaptic transmission in the developing visual cortex (1). ERK activation affects the axonal growth by phosphorylation of microtubule-associated proteins and neurofilaments (2).
Gene Aliases:
MAPK3; PRKM3; P44ERK1; P44MAPK; HS44KDAP; HUMKER1A; MGC20180
Genbank Number:
References:
1. Di Cristo, G. et.al: Requirement of ERK activation for visual cortical plasticity. Science 292: 2337-2340, 2001.
2. Forcet, C. et.al: Netrin-1-mediated axon outgrowth requires deleted in colorectal cancer-dependent MAPK activation. Nature 417: 443-447, 2002.
Specific Activity:
Sample Kinase Activity Plot. For specific information on a given lot, see related technical data sheet.
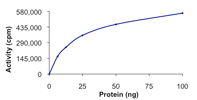
|
Purity:
Sample Purity Data. For specific information on a given lot, see related technical data sheet.
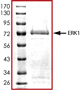
|
Storage, Stability and Shipping:
Store product at –70oC. For optimal storage, aliquot target into smaller quantities after centrifugation and store at recommended temperature. For most favorable performance, avoid repeated handling and multiple freeze/thaw cycles.
Molecular Weight:
~72 kDa
![]() Tanaka H et al., The intellectual disability gene PQBP1 rescues Alzheimer’s disease pathology Molecular Psychiatry October 2018 10.1038/s41380-018-0253-8
Tanaka H et al., The intellectual disability gene PQBP1 rescues Alzheimer’s disease pathology Molecular Psychiatry October 2018 10.1038/s41380-018-0253-8
![]() Liu F et al., Ethyl gallate as a novel ERK1/2 inhibitor suppresses patient-derived esophageal tumor growth Molecular Carcinogenesis December 2018 10.1002/mc.22948
Liu F et al., Ethyl gallate as a novel ERK1/2 inhibitor suppresses patient-derived esophageal tumor growth Molecular Carcinogenesis December 2018 10.1002/mc.22948
![]() Li Saiqun et al., RAS/ERK Signaling Controls Proneural Genetic Programs in Cortical Development and Gliomagenesis Journal of Neuroscience February 2014 10.1523/JNEUROSCI.4077-13.2014
Li Saiqun et al., RAS/ERK Signaling Controls Proneural Genetic Programs in Cortical Development and Gliomagenesis Journal of Neuroscience February 2014 10.1523/JNEUROSCI.4077-13.2014
![]() Tonga Junfeng et al., Phosphorylation of Rac1 T108 by Extracellular Signal-Regulated Kinase in Response to Epidermal Growth Factor: a Novel Mechanism To Regulate Rac1 Function Molecular and Cellular Biology November 2013 10.1128/MCB.00822-13
Tonga Junfeng et al., Phosphorylation of Rac1 T108 by Extracellular Signal-Regulated Kinase in Response to Epidermal Growth Factor: a Novel Mechanism To Regulate Rac1 Function Molecular and Cellular Biology November 2013 10.1128/MCB.00822-13
![]() E. Franklin Norah et al., Differential phosphorylation of the phosphoinositide 3-phosphatase MTMR2 regulates its association with early endosomal subtypes Journal of Cell Science February 2013 10.1242/jcs.113928
E. Franklin Norah et al., Differential phosphorylation of the phosphoinositide 3-phosphatase MTMR2 regulates its association with early endosomal subtypes Journal of Cell Science February 2013 10.1242/jcs.113928
![]() Y. Caro-Gonzalez2 Hector et al., Mitogen-activated protein kinase (MAPK/ERK) regulates adenomatous polyposis coli during growth-factor-induced cell extension Journal of Cell Science March 2012 10.1242/jcs.095166
Y. Caro-Gonzalez2 Hector et al., Mitogen-activated protein kinase (MAPK/ERK) regulates adenomatous polyposis coli during growth-factor-induced cell extension Journal of Cell Science March 2012 10.1242/jcs.095166
![]() Tyler Moore et al., IRF3 and ERK MAP-kinases control nitric oxide production from macrophages in response to poly-I:C FEBS Letters September 2013 10.1016/j.febslet.2013.07.025
Tyler Moore et al., IRF3 and ERK MAP-kinases control nitric oxide production from macrophages in response to poly-I:C FEBS Letters September 2013 10.1016/j.febslet.2013.07.025
![]() Yanga Shuping et al., Phosphorylation of KIBRA by the extracellular signal-regulated kinase (ERK)?ribosomal S6 kinase (RSK) cascade modulates cell proliferation and migration Cellular Signaling February 2014 10.1016/j.cellsig.2013.11.012
Yanga Shuping et al., Phosphorylation of KIBRA by the extracellular signal-regulated kinase (ERK)?ribosomal S6 kinase (RSK) cascade modulates cell proliferation and migration Cellular Signaling February 2014 10.1016/j.cellsig.2013.11.012
![]() Neise Denise et al., Evidence for a differential modulation of p53-phosphorylating kinases by the cyclin-dependent kinase inhibitor p21WAFI/C1P1 Cell Cycle September 2010 10.4161/cc.9.17.12799
Neise Denise et al., Evidence for a differential modulation of p53-phosphorylating kinases by the cyclin-dependent kinase inhibitor p21WAFI/C1P1 Cell Cycle September 2010 10.4161/cc.9.17.12799
![]() AL Lasek et al., The Functional Significance of Posttranslational Modifications on Polo-Like Kinase 1 Revealed by Chemical Genetic Complementation. PLoS One February 2016 10.1371/journal.pone.0150225
AL Lasek et al., The Functional Significance of Posttranslational Modifications on Polo-Like Kinase 1 Revealed by Chemical Genetic Complementation. PLoS One February 2016 10.1371/journal.pone.0150225
![]() Tong Junfeng et al., Phosphorylation and Activation of RhoA by ERK in Response to Epidermal Growth Factor Stimulation PLoS One January 2016 10.1371/journal.pone.0147103
Tong Junfeng et al., Phosphorylation and Activation of RhoA by ERK in Response to Epidermal Growth Factor Stimulation PLoS One January 2016 10.1371/journal.pone.0147103
![]() N Sato et al., MEK and PI3K catalytic activity as predictor of the response to molecularity targeted agents in triple-negative breast cancer Biochemical and Biophysical Research Communications August 2017
N Sato et al., MEK and PI3K catalytic activity as predictor of the response to molecularity targeted agents in triple-negative breast cancer Biochemical and Biophysical Research Communications August 2017
![]() Qiu Chen Jin et al., Absolute quantitation of endogenous proteins with precision and accuracy using a capillary Western system Analytical Biochemistry November 2013 10.1016/j.ab.2013.07.022
Qiu Chen Jin et al., Absolute quantitation of endogenous proteins with precision and accuracy using a capillary Western system Analytical Biochemistry November 2013 10.1016/j.ab.2013.07.022
![]() Bhandaria Deepali et al., Cyclin-dependent kinase 5 activates guanine nucleotide exchange factor GIV/Girdin to orchestrate migration?proliferation dichotomy PNAS July 2015 10.1073/pnas.1514157112
Bhandaria Deepali et al., Cyclin-dependent kinase 5 activates guanine nucleotide exchange factor GIV/Girdin to orchestrate migration?proliferation dichotomy PNAS July 2015 10.1073/pnas.1514157112
![]() K Fujita et al., HMGB1, a pathogenic molecule that induces neurite degeneration via TLR4-MARCKS, is a potential therapeutic target for Alzheimer's disease. Science Reports August 2016 10.1038/srep31895
K Fujita et al., HMGB1, a pathogenic molecule that induces neurite degeneration via TLR4-MARCKS, is a potential therapeutic target for Alzheimer's disease. Science Reports August 2016 10.1038/srep31895
![]() et al., Calmodulin Directly Interacts with the Cx43 Carboxyl-Terminus and Cytoplasmic Loop Containing Three ODDD-Linked Mutants (M147T, R148Q, and T154A) that Retain ?-Helical Structure, but Exhibit Loss-of-Function and Cellular Trafficking Defects Biomolecules October 2020
et al., Calmodulin Directly Interacts with the Cx43 Carboxyl-Terminus and Cytoplasmic Loop Containing Three ODDD-Linked Mutants (M147T, R148Q, and T154A) that Retain ?-Helical Structure, but Exhibit Loss-of-Function and Cellular Trafficking Defects Biomolecules October 2020
![]() et al., PPM1A Controls Diabetic Gene Programming through Directly Dephosphorylating PPAR? at Ser273 Cells February 2020
et al., PPM1A Controls Diabetic Gene Programming through Directly Dephosphorylating PPAR? at Ser273 Cells February 2020
![]() et al., Cell growth inhibition by 3-deoxysappanchalcone is mediated by directly targeting the TOPK signaling pathway in colon cancer. Phytomedicine : international journal of phytotherapy and phytopharmacology December 2019
et al., Cell growth inhibition by 3-deoxysappanchalcone is mediated by directly targeting the TOPK signaling pathway in colon cancer. Phytomedicine : international journal of phytotherapy and phytopharmacology December 2019
Angiogenesis, Apoptosis/Autophagy, Cancer, Cardiovascular Disease, ERK/MAPK Pathway, Invasion/Metastasis, Neurobiology, Ser/Thr Kinases
STAY CONNECTED
Fax: 1-604-232-4601